Houseplants have always been a vital part of peoples’ lives worldwide. There’s something super satisfying about seeing your plants thriving under your care. Every new growth, bloom, and propagation sparks joy for so many hobbyists. I’ve noticed an explosion in the number of people who have become houseplant hobbyists in recent years, probably due to pandemic-related quarantines and people spending more time in the comfort of their own homes.
Trial and error is involved in a lot of aspects of plant care, such as figuring out the required lighting, temperature, watering schedule, and humidity. I know from experience that a lot of houseplants manage to make it through rough patches and are quite forgiving when it comes to your learning curve!
One thing that you’re better off understanding right off the bat without any trial and error is plant toxicity. I’m sure many houseplant owners, newbies and veterans alike, have considered plant toxicity and how careful they should be when handling or owning certain plants. Especially in the context of children or pets, this is a really important aspect of the houseplant hobby to understand. The main culprit we read about is “calcium oxalate”.
Oxalic Acid
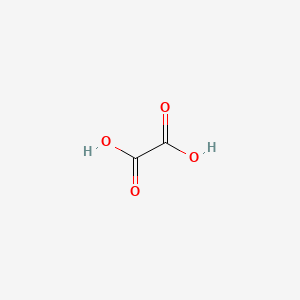
Let’s first discuss oxalic acid. Oxalic acid has the chemical formula C2H2O4. When the two hydrogen ions come off, there’s an overall 2- charge on C2O4, which is oxalate, the anion (negatively charged) form of that molecule. Sodium, potassium, calcium, and even magnesium can join with the oxalate and form compounds such as sodium oxalate, potassium oxalate, calcium oxalate, and magnesium oxalate. These are the types of compounds that we’ll discuss today!
Oxalates themselves are found in over 1000 different plant genera1, and not all of those plant genera are “houseplants” – many of them are actually foods that we eat (more on that below). Oxalates come in 2 forms: insoluble and soluble. In a nutshell, the insoluble calcium oxalate is the toxic type found in houseplants, and the soluble form is found in a lot of our foods, and is generally okay to eat if you’re healthy and it’s at low levels.
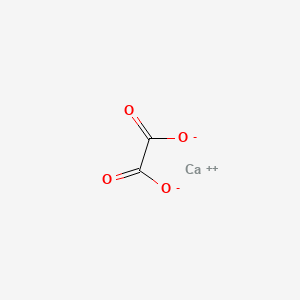
Insoluble calcium oxalate
Generally, the insoluble calcium oxalate crystals are the type found in our beloved houseplants. Here are some of the most popular plant genera that have been identified to contain moderate to high levels of insoluble calcium oxalate:
- Spathiphyllum (Peace lily)
- Alocasia (African mask)
- Colocasia (Elephant ear)
- Monstera (Swiss cheese)
- Caladium
- Epipremnum (Pothos, Devil’s ivy)
- Philodendron
- Diffenbachia (Dumb cane)
- Zantedeschia (Calla lily)
These plants form calcium oxalate crystals in order to regulate their own calcium levels; the crystals act as a calcium reserve.1 They also function as protection against predation. These needle-sharp crystals usually exist in formations called raphides. When an animal bites into the plant tissue, the crystals are released and cause irritation, swelling of the oropharyngeal region, and vomiting.2 It’s recommended by the ASPCA to keep these types of plants away from pets. They have a great, comprehensive list for cats and dogs. A popular recommendation is to provide cats with cat grass instead.
Soluble oxalates
Soluble oxalates exist in the form of soluble salts, such as sodium oxalate and potassium oxalate. “Soluble salts” refers to the fact that the compound is generally found separated into the salt ion (such as 2 Na+) and the oxalate ion (C2O42-), in water.

Many of the foods we eat, such as spinach3, star fruit4, beets5, quinoa5, peanuts6, and rhubarb7 contain soluble oxalates. Many analytical studies have measured the levels of soluble oxalates in these foods before and after various preparation techniques. People with healthy gut and kidney function are generally fine eating these foods. Low levels of soluble oxalates and insoluble calcium oxalate crystals (that form in our bodies if the soluble oxalate combines with calcium in our bodies) easily pass through the gut and bloodstream and end up excreted, or are simply degraded by the bacteria in our gut, such as Oxalobacter formigenes and Lactobacillus.8 People who are dehydrated, are on dialysis, or have kidney stones are generally advised to avoid foods that are high in oxalates.
Conclusion
In summary, oxalates are molecules that are found in all sorts of plants, not just a handful of well-known houseplants. They can be found in the form of insoluble calcium oxalate crystals in some of our beloved houseplants, and ingestion can hurt pets and humans alike, so it’s best to keep these types of houseplants out of our mouths. Oxalates are found in our foods, too, but no need to change up your meal plans for this reason alone, as foods that contain oxalates are also very nutritious. Healthy folks are generally fine ingesting the levels of oxalates found in foods, and clear it easily.
